News
Nature Communications publishes paper first-authored by JI student
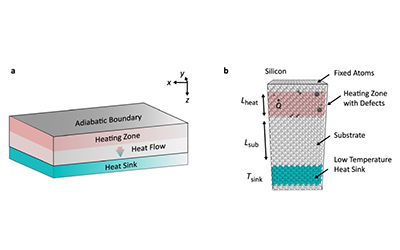
Yue Hu, a Class of 2023 doctoral graduate of the University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter), has published a research paper titled “Defect scattering can lead to enhanced phonon transport at the nanoscale” as the first author in the esteemed academic journal Nature Communications.
Embracing diversity: a JI journey with Niladri Saha Saccha
Editor’s Note: Niladri Saha Saccha, a Bangladeshi student, embarked on a transformative journey at the University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) in Fall 2021. As he approaches the culmination of his undergraduate...
Two JI professors rank among 2023 Most Cited Chinese Researchers
Professor Peisen Huang and Professor Xudong Wang of the University of Michigan – Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) have been listed as 2023 Most Cited Chinese Researchers by Elsevier, a Netherlands-based academic publishing...
JI launches Trip.com scholarship to foster cross-cultural understanding
The University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) has joined hands with Trip.com Group, a leading global travel service provider, to launch a scholarship exchange program to foster cross-cultural understanding and...
JI celebrates 18th anniversary with alumni homecoming event
The University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) celebrated its 18th anniversary and hosted an alumni homecoming event at Long Bin Building on April 6, coinciding with the 128th anniversary of SJTU.
JI holds 2024 graduate commencement ceremony
The 2024 University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) Graduate Commencement Ceremony was held at Long Bin Building on March 29.
JI and GIFT unite in 2024 Spring Fun Sports Games
The University of Michigan- Shanghai Jiao Tong University Joint Institute (UM-SJTU JI,JI hereafter) joined hands with SJTU’s Global Institute of Future Technology (GIFT) to host a vibrant and energetic 2024 Spring Fun Sports Games recently, bringing together over 300 students, faculty and staff members in a celebration of unity and achievement.
JI professor’s PNAS paper advances catalyst design optimization
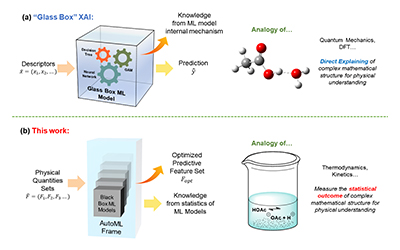
The research team led by Assistant Professor Yulian He of the University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) has made a significant contribution in catalyst design optimization. Their findings, published in the prestigious scientific journal, Proceedings of the National Academy of Sciences of the United States of America (PNAS), introduce a novel method for determining the pivotal physical quantity influencing chemisorption energy on catalyst surfaces.
The Trip.com Group Sponsored Scholarship Exchange Program is Now Open for Applications
To foster cultural and educational exchanges between the two countries, an agreement has been reached between China and the US to increase the number of American students studying in China. To support this goal, the UM-SJTU Joint Institute (the Joint Institute) is...
JI winter study abroad programs revitalize with high attendance
More than 280 students of the University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) have joined the winter study abroad programs organized by the JI International Programs Office over the past two months.
JI receives UM delegation led by President Santa Ono
The University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) warmly received UM President Santa Ono and his delegation on February 3, marking the president’s inaugural on-site visit to the SJTU campus since assuming office in October 2022.
JI students excel at 2023 University Physics Competition
The University of Michigan-Shanghai Jiao Tong University Joint Institute (UM-SJTU JI, JI hereafter) students showcased excellent performance in the 2023 University Physics Competition, securing four gold medals, 15 silver medals, and 14 bronze medals as key members of...